Nucleus atom10/31/2022  /atomic-structure-artwork-549603139-57fe40e75f9b586c3537ebf4.jpg)
The atomic number of an α-particle is Z=2, so the atomic number of the decaying nucleus is decreased by two during an α-decay and a different element is created. An α-particle is the nucleus of a helium atom, helium-4, which consists of two protons and two neutrons. When an atomic nucleus transforms into a different element by emitting an α-particle. 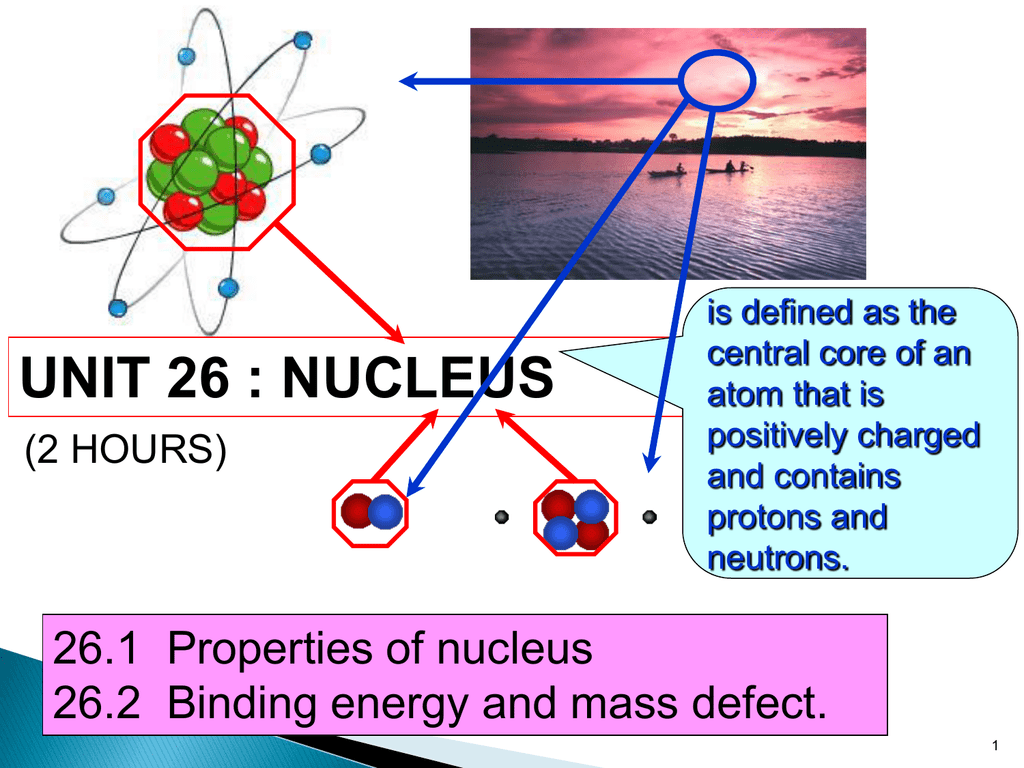
In 1899 Ernest Rutherford named alpha (α), beta (β), and gamma (γ) radiation, after the first three letters of the Greek alphabet. The different types of radiation can be identified by their ability to pass through matter. Radioactivity is the spontaneous decay of an unstable atom through the emission, from the atomic nucleus, of a particle of ionising radiation. 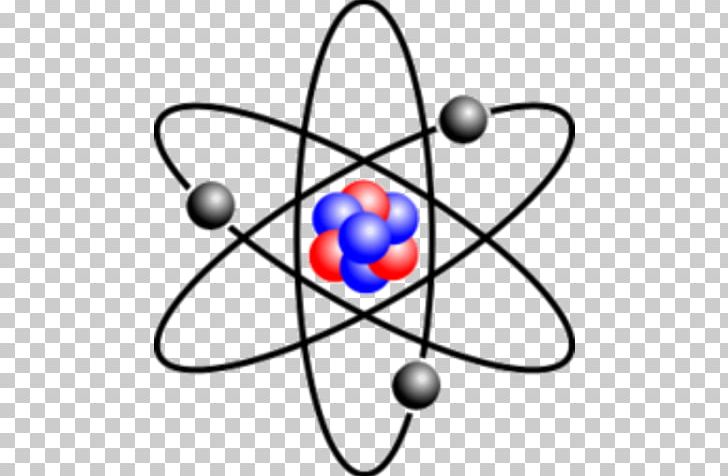
There are less than 300 stable nuclei and over 3,000 unstable radioactive nuclei.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
